A high incidence of pulmonary embolism has been described during the coronavirus pandemic.
MethodsThis work is a single-center retrospective study which reviewed computed tomography pulmonary angiograms ordered due to suspected pulmonary embolism during two periods: from March 1, 2020 to May 31, 2020 (pandemic) and during the same interval in 2019 (control).
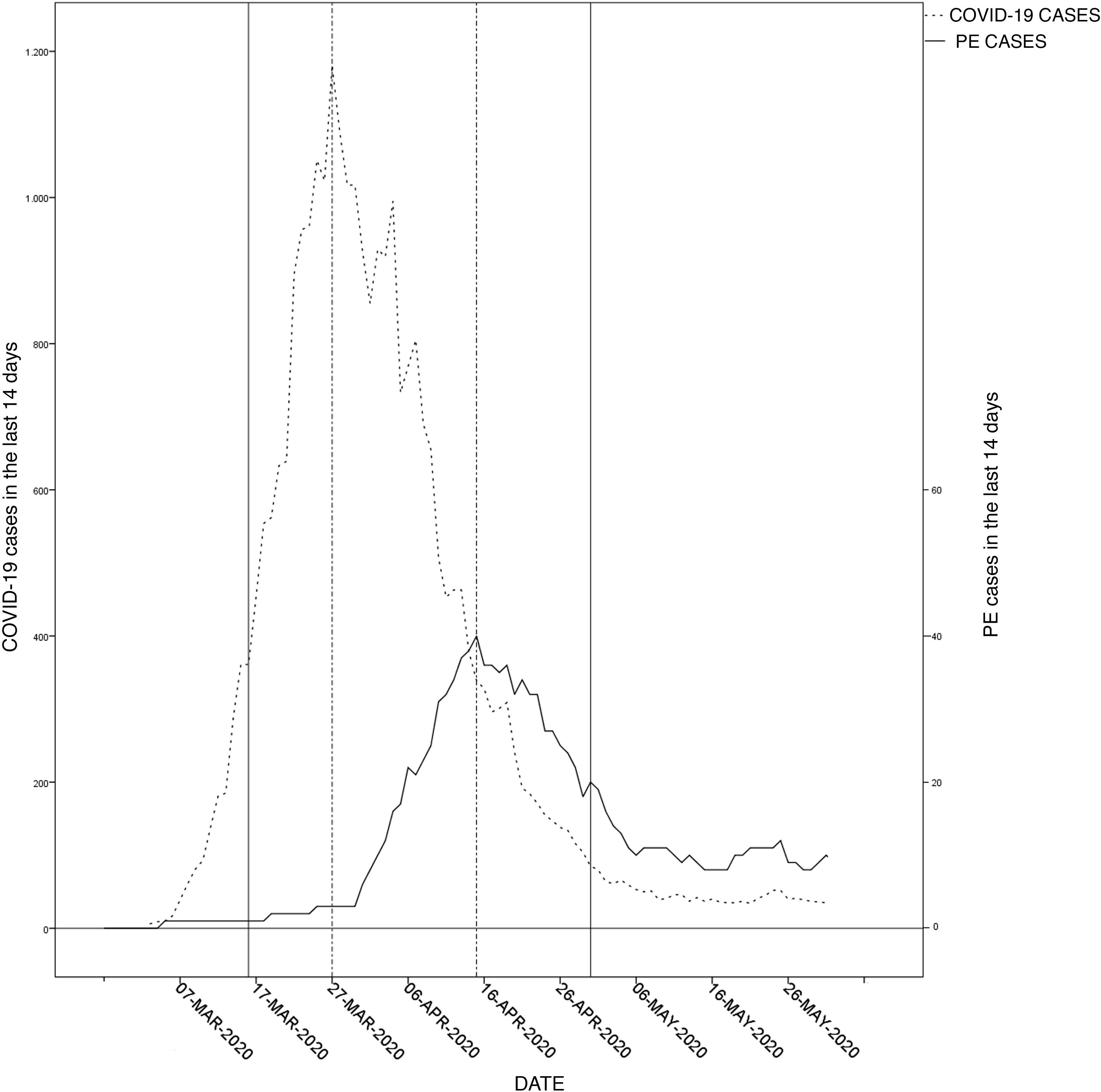
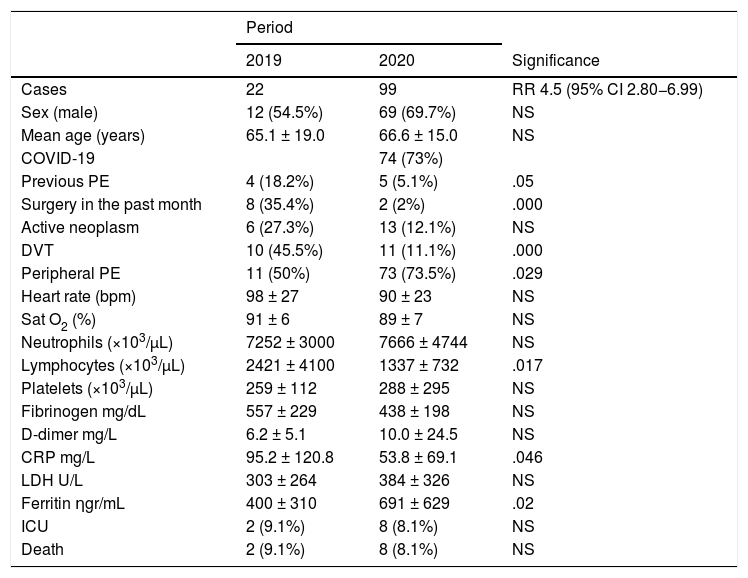
ResultsTwenty-two pulmonary embolisms were diagnosed during the control period and 99 in the pandemic, 74 of which were associated with COVID-19. Of all patients hospitalized with COVID-19, 5.3% had a pulmonary embolism, with a delay between the two diagnoses of 9.1 ± 8.4 days. During the pandemic, patients with pulmonary embolism had fewer predisposing conditions (previous pulmonary embolism 5.1 vs. 18.2%, p = .05; previous surgery 2 vs. 35.4%, p = .0001; deep vein thrombosis 11.1 vs. 45.5%, p = .0001); peripheral pulmonary embolisms were the most frequent (73.5 vs. 50%, p = . 029).
ConclusionsThere is an increased risk of having a pulmonary embolism during the SARS-CoV-2 pandemic, which affects patients with a different clinical profile and more often causes distal pulmonary embolisms.
Se ha descrito una elevada incidencia de tromboembolismo pulmonar (TEP) durante la pandemia por coronavirus.
MétodosEstudio retrospectivo unicéntrico, con revisión de las angiografías pulmonares por tomografía computarizada solicitadas por sospecha de tromboembolismo pulmonar durante dos períodos, del 01 de marzo del 2020 al 31de mayo del 2020 (pandemia), e igual intervalo en 2019 (control).
ResultadosSe diagnosticaron 22 tromboembolismos pulmonares durante el período control y 99 en el pandémico, 74 asociados con COVID-19. El 5,3% de los pacientes hospitalizados con COVID-19 sufrió un tromboembolismo pulmonar, con un retraso entre ambos diagnósticos de 9,1 ± 8,4 días. Durante la pandemia, los pacientes con tromboembolismo pulmonar tenían menos condiciones predisponentes (tromboembolismo pulmonar previo 5,1 vs. 18,2%, p = 0,05, cirugía previa 2 vs. 35,4%, p = 0,0001, trombosis venosa profunda 11,1 vs. 45,5%, p = 0,0001), y los tromboembolismos pulmonares periféricos eran más frecuentes (73,5 vs. 50%, p = 0,029).
ConclusionesExiste un riesgo incrementado de sufrir un TEP durante la pandemia por SARS-CoV-2, que afecta a pacientes con perfil clínico diferente y causa más frecuentemente TEP distales.
Article
Diríjase desde aquí a la web de la >>>FESEMI<<< e inicie sesión mediante el formulario que se encuentra en la barra superior, pulsando sobre el candado.

Una vez autentificado, en la misma web de FESEMI, en el menú superior, elija la opción deseada.

>>>FESEMI<<<