A new coronavirus disease in humans, COVID-19, caused by SARS-CoV-2, emerged in December 2019. It has been associated with the development of thrombotic phenomena. Retinal vein occlusion (RVO) is mainly a consequence of vascular risk factors (VRF). This study aimed to analyze cases of COVID-19 in a cohort of patients with RVO (Valdecilla cohort).
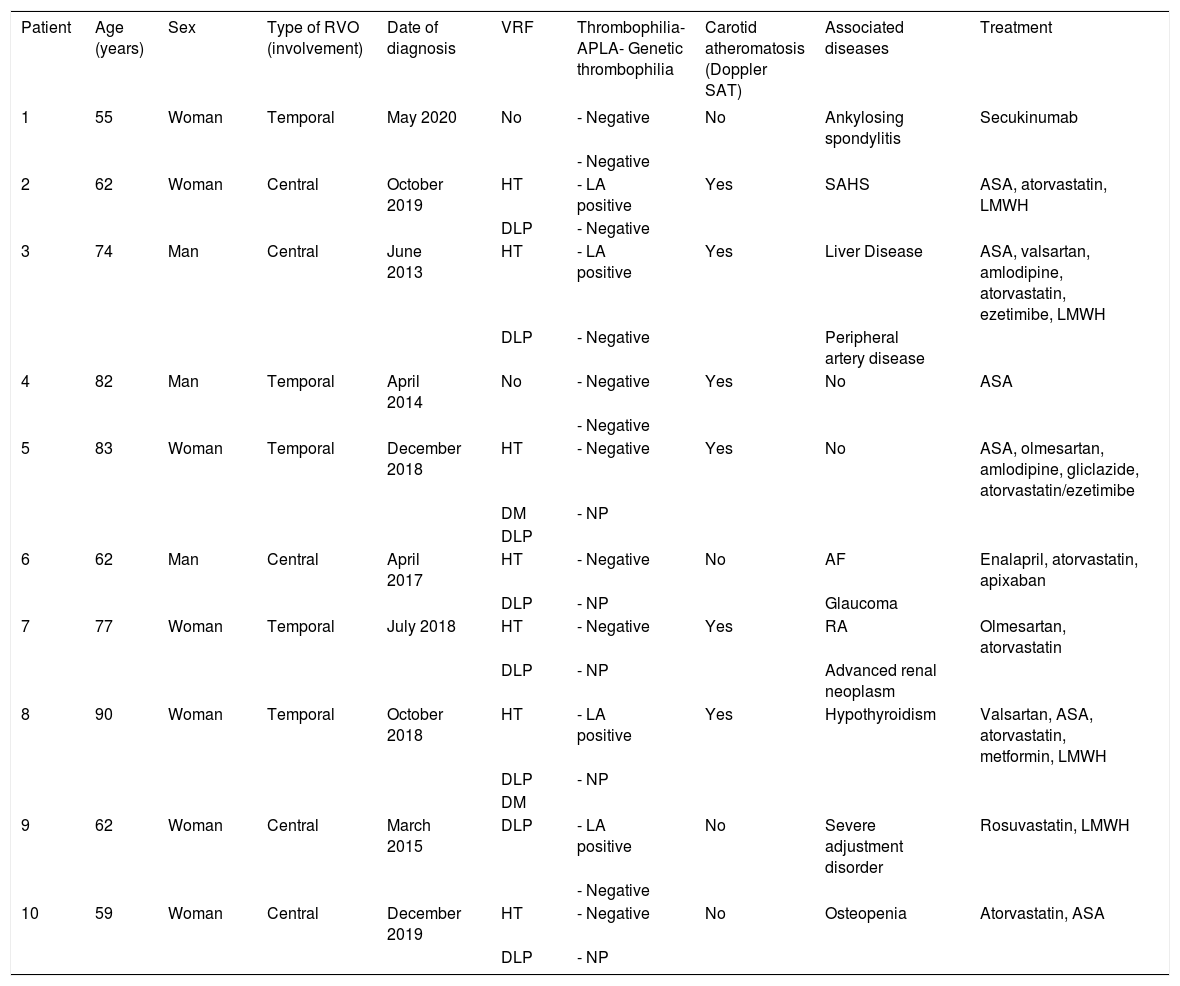
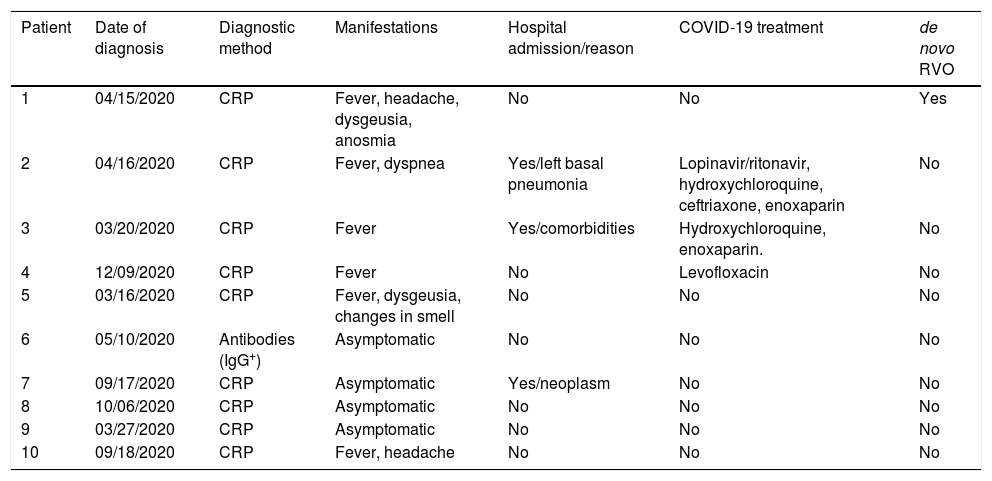
Patients and methodsBetween December 2008 and December 2020, 429 patients with RVO were attended to in our clinic. Ten patients had COVID-19, one of which did not have VRF or thrombophilia. The remaining nine patients had RVO prior to the infection and VRF, six had carotid atherosclerosis, and four had antiphospholipid syndrome. The infection did not cause thrombotic phenomena in any of them.
ConclusionsRVO is a rare manifestation of COVID-19. In our cohort of patients with RVO, COVID-19 disease did not lead to thrombotic events.
En diciembre de 2019 surgió una nueva enfermedad por coronavirus en humanos causada por el virus SARS-CoV-2, la COVID-19, que se ha asociado con fenómenos trombóticos. La obstrucción venosa retiniana (OVR) es principalmente una consecuencia de los factores de riesgo vascular (FRV). El objetivo de este estudio ha sido analizar los casos de infección por SARS-CoV-2 en una cohorte de pacientes con OVR (cohorte Valdecilla).
Pacientes y métodosEntre diciembre de 2008 y 2020 hemos atendido 429 pacientes con OVR. Diez han padecido COVID-19, de los que uno no presentaba FRV ni trombofilia. Los otros 9 fueron diagnosticados de OVR antes de la infección: todos tenían FRV; 6 ateromatosis carotídea y 4 presentaban un síndrome antifosfolípido. La infección no causó en ellos fenómenos trombóticos.
ConclusionesLa OVR es una manifestación infrecuente de la COVID-19. En nuestra cohorte de pacientes con OVR la COVID-19 no indujo eventos trombóticos.
Article
Diríjase desde aquí a la web de la >>>FESEMI<<< e inicie sesión mediante el formulario que se encuentra en la barra superior, pulsando sobre el candado.

Una vez autentificado, en la misma web de FESEMI, en el menú superior, elija la opción deseada.

>>>FESEMI<<<