The effect of dexamethasone in the initial phase of infection by SARS-CoV-2 and its influence on COVID-19 is not well defined. We describe clinical-radiological characteristics, the cytokine storm parameters, and the clinical evolution of a series of patients treated with dexamethasone in the disease's initial phase.
MethodA study of 8 patients who received dexamethasone before the development of COVID-19. We evaluate clinical variables, imaging tests, cytokine release parameters, treatment used and patient evolution.
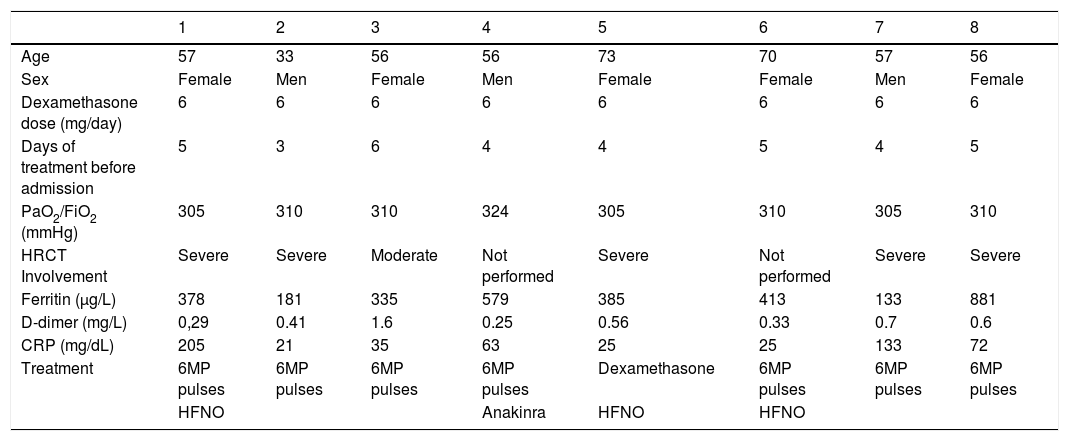
ResultsAll patients received a 6 mg/day dose with a mean duration of 4.5 days before admission. High resolution computed tomography (HRCT) revealed that most of them presented a severe extension; most patients had a slightly elevated level of cytokine release parameters. Three patients required high-flow oxygen therapy due to respiratory failure; none required orotracheal intubation or died.
ConclusionDexamethasone in the early stages of SARS-CoV-2 infection appears to be associated with severe COVID-19.
El efecto de la dexametasona en la fase inicial de la infección por SARS-CoV-2 y su influencia sobre la COVID-19 no está bien definido. Describimos las características clínico-radiológicas, los parámetros de tormenta de citoquinas y la evolución clínica de una serie de pacientes tratados con dexametasona en la fase inicial de la enfermedad.
MétodoEstudio de 8 pacientes que recibieron dexametasona previo al desarrollo de la COVID-19. Evaluamos variables clínicas, pruebas de imagen, parámetros de liberación de citoquinas, el tratamiento empleado y su evolución.
ResultadosTodos los pacientes recibieron una dosis de 6 mg/día con una duración media de 4,5 días previos al ingreso. La mayoría de los pacientes presentaron una extensión grave en la tomografía computarizada de alta resolución (TCAR) y una elevación leve de los parámetros de liberación de citoquinas; tres pacientes requirieron oxigenoterpia nasal de alto flujo (ONAF) por insuficiencia respiratoria, y ningún paciente requirió intubación orotraqueal ni falleció.
ConclusiónLa dexametasona en las fases iniciales de la infección por SARS-CoV-2 parece asociarse con una COVID-19 grave.
Article
Diríjase desde aquí a la web de la >>>FESEMI<<< e inicie sesión mediante el formulario que se encuentra en la barra superior, pulsando sobre el candado.

Una vez autentificado, en la misma web de FESEMI, en el menú superior, elija la opción deseada.

>>>FESEMI<<<