The SERPINA1 gene encodes the protein Alpha-1 Antitrypsin (AAT1). Possible imbalances between the concentrations of proteases and antiproteases (AAT1) can lead to the development of serious pulmonary and extrapulmonary pathologies. In this work we study the importance of this possible imbalance in patients with COVID-19.
ObjectivesTo correlate the severity of the symptoms of SARS-COV-2 infection with the AAT1 concentrations at diagnosis of the disease.
MethodsAn observational, prospective, cross-sectional, non-interventional, analytical study was carried out where 181 cases with COVID-19 admitted to the “Lozano Blesa” University Clinical Hospital of Zaragoza were selected. The concentration of AAT1 was studied in all of them and this was correlated with the clinical aspects and biochemical parameters at hospital admission.
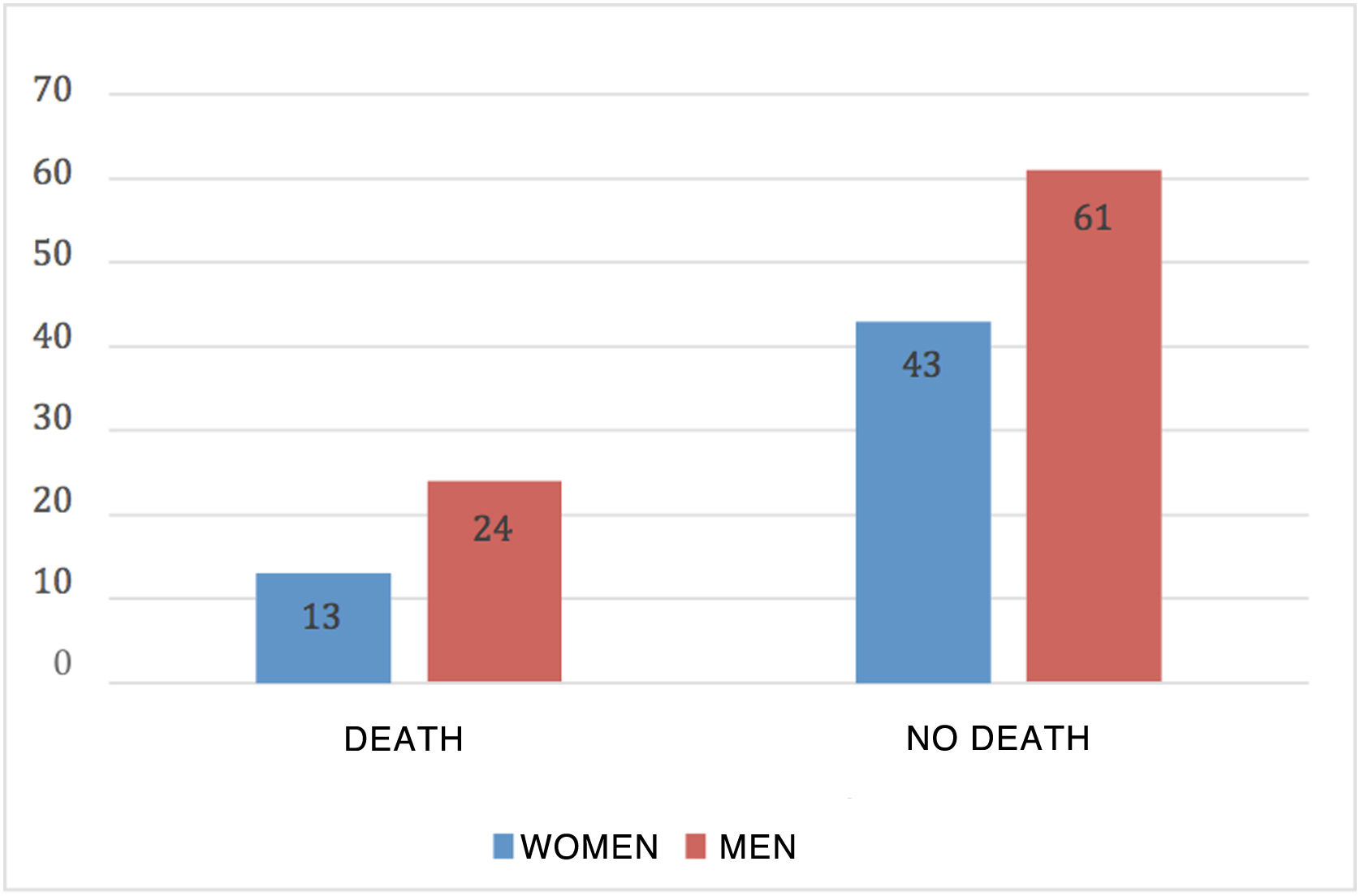
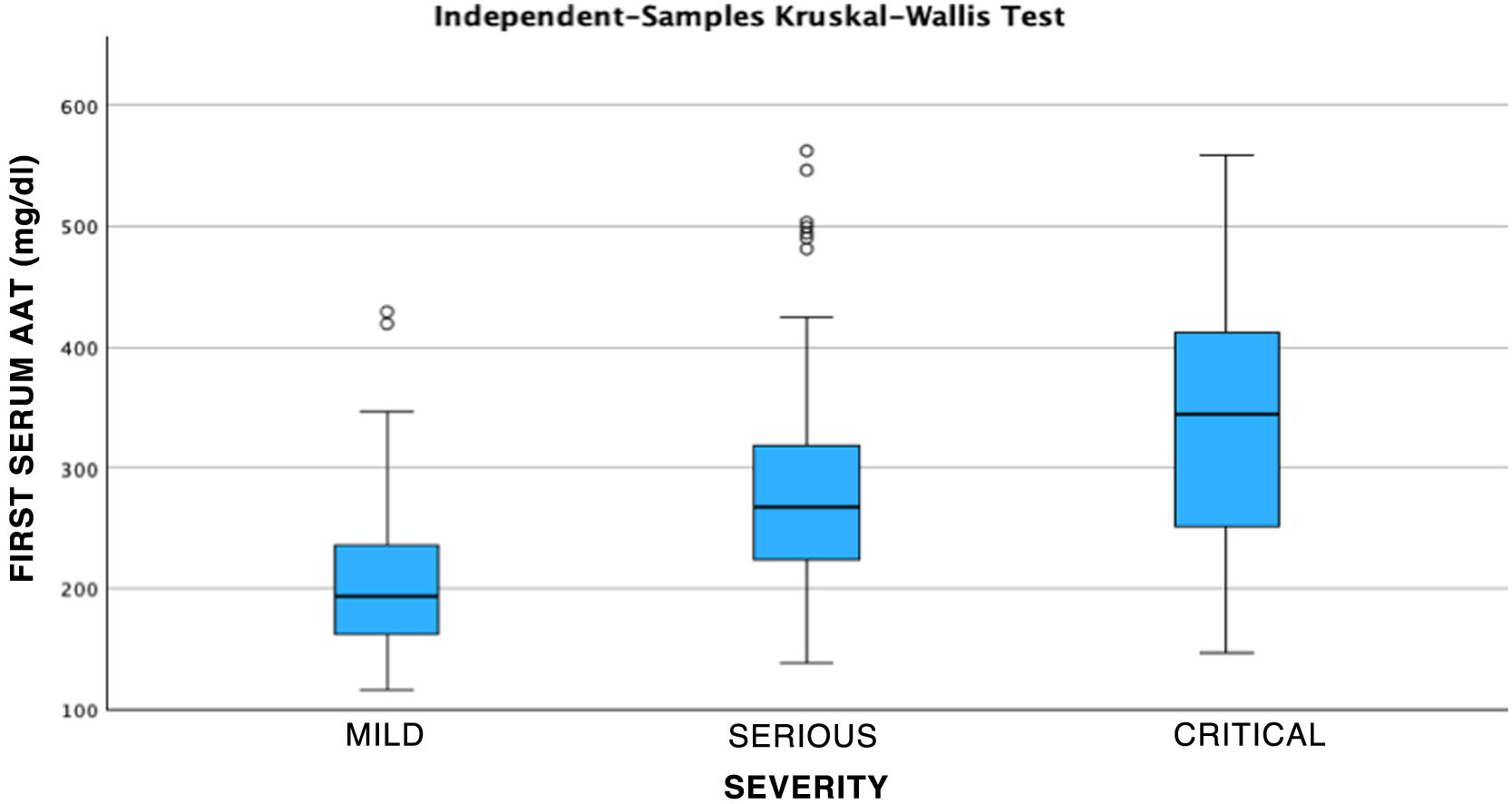
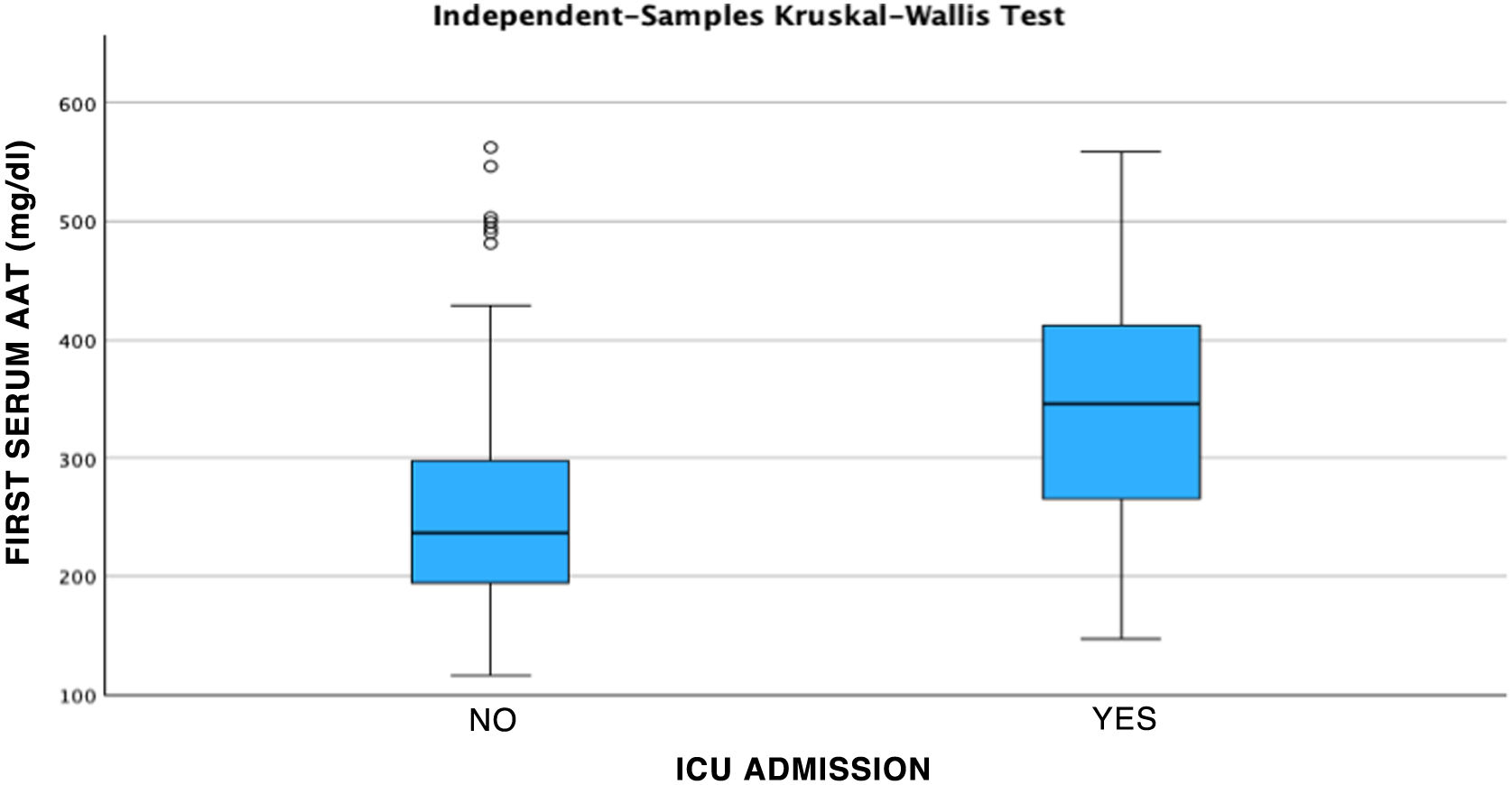
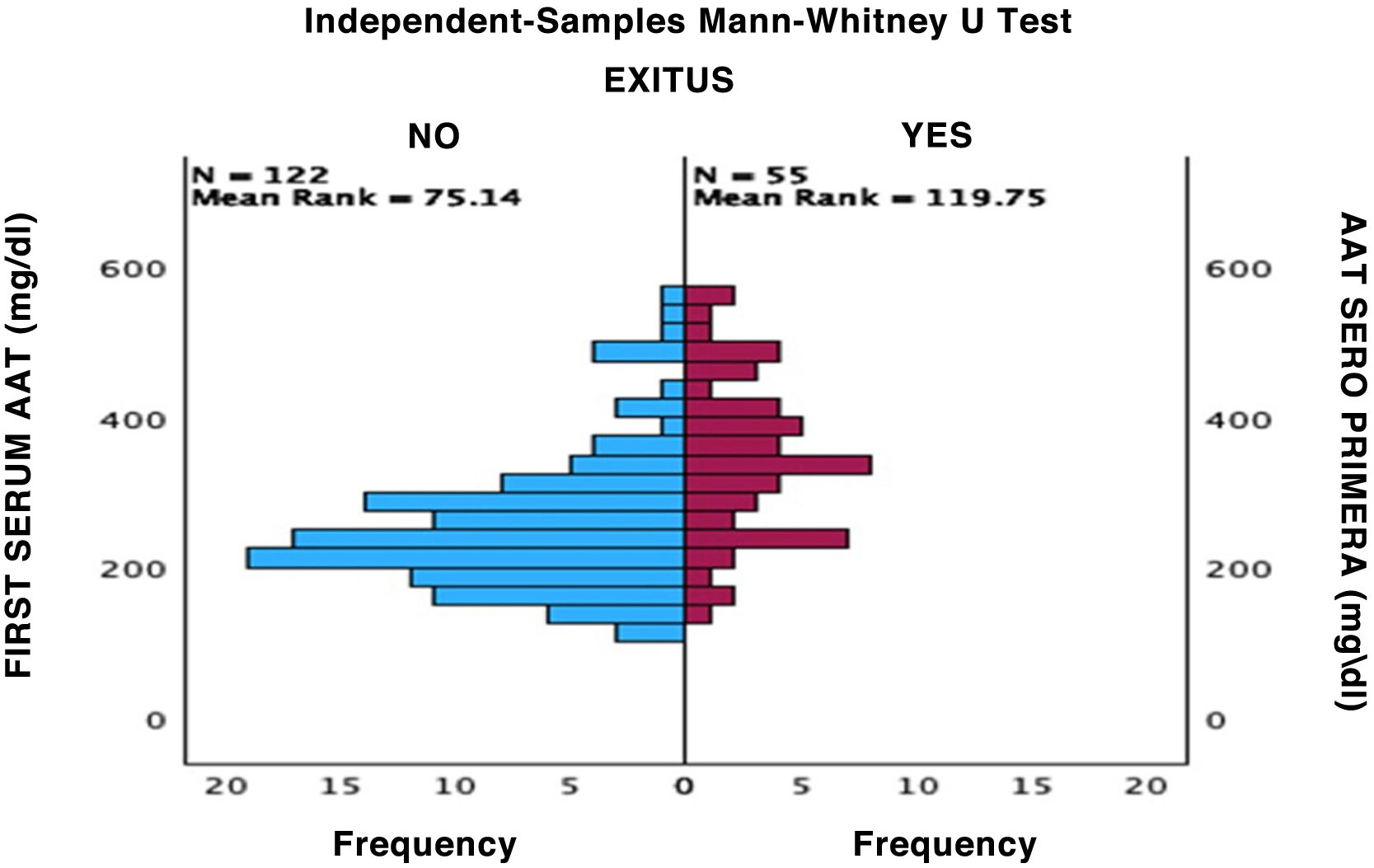
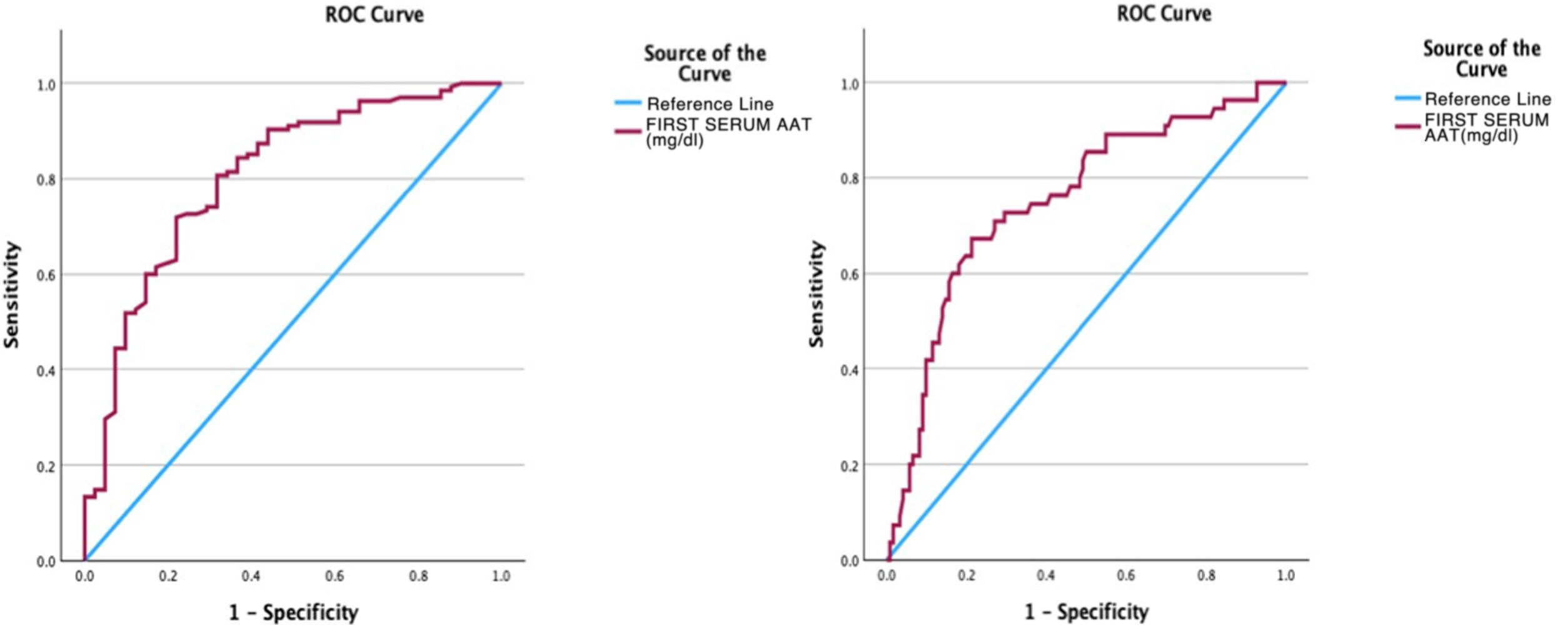
Results141 cases corresponded to patients with severe COVID and 40 patients with mild COVID. AAT1 levels were positively correlated with the days of hospitalization, severity, C-Reactive Protein, ferritin, admission to Intensive Care, and death, and presented a negative correlation with the number of lymphocytes/mm3. AAT1 concentrations higher than 237.5 mg/dL allowed the patient to be classified as “severe” (S72%; E78%) and 311.5 mg/dL were associated with the risk of admission to Intensive Care or Exitus (S67%; E79%).
ConclusionsLevels of the SERPINA1 gene expression product, AAT1, correlate with the severity of COVID-19 patients at diagnosis of the disease, being useful as a prognostic biomarker.
El gen SERPINA1 codifica la proteína Alfa-1 Antitripsina (AAT1). Los posibles desequilibrios entre las concentraciones de proteasas y antiproteasas (AAT1) puede hacer que se desarrollen patologías pulmonares y extrapulmonares graves. En este trabajo estudiamos la importancia de este posible disbalance en los pacientes con COVID-19.
ObjetivosCorrelacionar la gravedad de la clínica de la infección por SARS-COV-2 con las concentraciones de AAT1 al diagnóstico de la enfermedad.
MétodosSe ha llevado a cabo un estudio observacional, prospectivo, transversal, no intervencionista con carácter analítico donde se seleccionaron 181 casos con COVID-19 ingresados en el Hospital Clínico Universitario “Lozano Blesa” de Zaragoza. A todos ellos se les estudió la concentración de AAT1 y ésta se correlacionó con los aspectos clínicos y parámetros bioquímicos al ingreso hospitalario.
Resultados141 casos correspondían a pacientes con COVID grave y 40 pacientes con COVID leve. Los niveles de AAT1 se correlacionaron positivamente con los días de estancia, la gravedad, la Proteína C Reactiva, la ferritina, el ingreso en Cuidados Intensivos y el fallecimiento y presentaron una correlación negativa con el número de linfocitos/mm3. Las concentraciones de AAT1 superiores a 237,5 mg/dL permitieron catalogar al paciente como “grave” (S72%; E78%) y de 311,5 mg/dL se asociaron con el riesgo de ingreso en Cuidados Intensivos o Exitus (S67%; E79%).
ConclusionesLos niveles del producto de la expresión del gen SERPINA1, la AAT1, se correlacionan con la gravedad de los pacientes COVID-19 al diagnóstico de la enfermedad siendo útil como biomarcador pronóstico.
Article
Diríjase desde aquí a la web de la >>>FESEMI<<< e inicie sesión mediante el formulario que se encuentra en la barra superior, pulsando sobre el candado.

Una vez autentificado, en la misma web de FESEMI, en el menú superior, elija la opción deseada.

>>>FESEMI<<<