This study aimed to evaluate the performance, before and during the COVID-19 pandemic, of the case definition of suspected influenza used in community surveillance in Mexico.
MethodsA cross-sectional analysis of a cohort study was performed and cases that met the suspected case criteria (n = 20,511) and that had laboratory-conclusive evidence (quantitative real-time polymerase chain reaction) to confirm or discard influenza virus infection were analysed.
ResultsA high sensitivity and modest specificity were documented, which later decreased during the COVID-19 outbreak, as well as its diagnostic accuracy. However, no significant differences were observed in the area under the receiver operating characteristics curve among the analysed periods.
ConclusionsThe evaluated case definition remains to be a cost-effective alternative for identifying patients who may benefit from influenza-specific antiviral drugs, even during the global COVID-19 outbreak.
Este estudio tuvo como objetivo evaluar el desempeño, antes y durante la pandemia de la COVID-19, de la definición de caso de sospecha de influenza en la vigilancia comunitaria en México.
MétodosSe llevó a cabo un análisis transversal de un estudio de cohorte y, con el fin de confirmar o descartar la infección por virus influenza, se analizaron los casos que cumplían con los criterios de caso sospechoso (n = 20.511) y con evidencia concluyente de laboratorio (reacción cuantitativa en cadena de la polimerasa en tiempo real).
ResultadosSe documentaron una alta sensibilidad y una discreta especificidad, que posteriormente disminuyó, así como su precisión diagnóstica, durante el brote de la COVID-19. Sin embargo, no se observaron diferencias en el área bajo la curva de características operativas del receptor entre los períodos analizados.
ConclusionesLa definición de caso evaluada sigue siendo una alternativa rentable para identificar a los pacientes que pueden beneficiarse de los medicamentos antivirales específicos contra la influenza, incluso durante el brote mundial de COVID-19.
The influenza-related burden remains high globally despite vaccination efforts.1
In the northern hemisphere, seasonal cases start in October and tail off by May.2 The case definitions of suspected influenza used in national and regional surveillance programs commonly differ from those recommended by the World Health Organization (WHO).
In Mexico, according to regulatory standards,3 the case definition in patients aged 5 years or older includes the presence of fever (38 °C or higher), headache, and cough accompanied by at least 2 of the following symptoms: rhinorrhoea, coryza, arthritis, arthralgia, myalgia, exhaustion, odynophagia, thoracic pain, abdominal pain, nasal congestion or diarrhoea. Fever is not a cardinal symptom among older subjects (65+ years old).
Suspected cases that meet the criteria are classified as influenza-like illness (ILI) or severe acute respiratory infection (SARI) if systemic or worsening symptoms are present. This definition has several similarities to that proposed by the Groupes Régionaux d’Observation de la Grippe (GROG, according to the French acronym),4 which has shown good performance in community surveillance of influenza.5
The first recorded cases in Mexico of locally acquired coronavirus disease 2019 (COVID-19) due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) occurred in late February 2020.6 About three months later, over 75,000 cases and 8000 deaths had been recorded at the national level.7
Given that suspected cases of COVID-19 and influenza share clinical similarities,8 timely identification of the latter, which may benefit from the use of neuraminidase inhibitors (NAIs),9 can be challenging in healthcare settings with limitations.
We aimed to evaluate the performance of the influenza case definition before and during the COVID-19 pandemic in Mexico. We analysed two consecutive flu seasons (2018–2020) to benefit from a wider timeframe.
MethodsWe conducted a cross-sectional analysis of a nationwide retrospective cohort study. Suspected cases of influenza among individuals aged 5 years or older recorded during two consecutive seasons (2018–2020) in a regulatory system for the epidemiological surveillance of viral respiratory diseases (SISVER, according to the Spanish acronym), and which were later confirmed or ruled out as cases of influenza virus infection, were eligible.
Quantitative real-time polymerase chain reaction (qRT-PCR; SuperScript® III Platinum® One-step RT-qPCR System) analyses were performed on clinical specimens (nasopharyngeal or deep nasal swab). A detailed description of the laboratory methods used by the Mexican Institute of Social Security (IMSS, according to the Spanish acronym) network was previously published.10
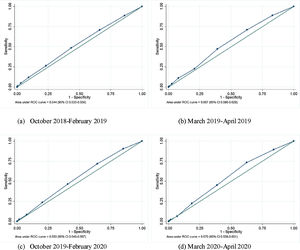
The performance of a case definition of suspected influenza was evaluated in terms of sensitivity, specificity, accuracy, and positive and negative likelihood ratio (LR+/−). Age (5–9; 10–19; 20–44; 45–64 and 65 years or older) and time-stratified (according to onset of symptoms: Oct. 2018–Feb. 2019; Mar. 2019–Apr. 2019; Oct. 2019–Feb. 2020; Mar. 2020–Apr. 2020) estimates were obtained. The fourth period was the pandemic period. The area under receiver operating characteristic curves (AUROCs) and 95% confidence intervals (CI) were also computed.
This study was approved by the Local Research Ethics Committee (601) of the IMSS (R-2020-601-015).
ResultsData from 20,511 cases were analysed. The overall prevalence of laboratory-confirmed influenza in the study sample was 38.8% (n = 7955). Table 1 summarizes the estimates. The prevalence of laboratory-positive influenza was lower among older subjects, particularly during the COVID-19 pandemic period (65+ years old, 13.2%). The mean sensitivity of case definition was high in all age groups and the last general estimate (92.7, 95% CI 91.3–94.1) was similar to the previous ones (p = 0.274).
Performance of suspected influenza case definition, Mexico 2018–2020.
| Age | n | Prevalence | Estimate (95% confidence interval) | LR+ | LR− | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sensitivity | Specificity | Accuracy | ||||||||
| Period 1 (October 2018–February 2019) | ||||||||||
| 5–9 | 626 | 59.3 | 91.1 | (88.9–93.3) | 16.5 | (13.6–19.4) | 60.7 | (56.9–64.5) | 1.091 | 0.540 |
| 10–19 | 539 | 49.5 | 92.1 | (89.9–94.4) | 12.9 | (10.0–15.7) | 52.1 | (47.9–56.3) | 1.057 | 0.611 |
| 20–44 | 4,159 | 38.6 | 94.1 | (93.4–94.8) | 8.6 | (7.8−9.5) | 41.6 | (40.1–43.1) | 1.029 | 0.688 |
| 45–64 | 2,806 | 33.8 | 91.1 | (90.1–92.2) | 9.5 | (8.4–10.6) | 37.1 | (35.3–38.8) | 1.007 | 0.932 |
| 65+ | 2,001 | 15.9 | 87.4 | (86.0–88.9) | 14.8 | (13.2–16.4) | 26.3 | (24.4–28.3) | 1.026 | 0.850 |
| All | 10,131 | 34.6 | 92.2 | (91.7–92.7) | 10.9 | (10.3–11.5) | 39.1 | (38.1–40.0) | 1.035 | 0.713 |
| Period 2 (March 2019–April 2019) | ||||||||||
| 5–9 | 214 | 65.4 | 92.9 | (89.4–96.3) | 13.5 | (8.9–18.1) | 65.4 | (59.1–71.8) | 1.074 | 0.529 |
| 10–19 | 224 | 63.0 | 93.6 | (90.4–96.8) | 10.8 | (6.8–14.9) | 63.0 | (56.6–69.3) | 1.050 | 0.589 |
| 20–44 | 841 | 40.8 | 96.8 | (95.6–98.0) | 9.4 | (7.5–11.4) | 45.1 | (41.7–48.4) | 1.069 | 0.340 |
| 45−64 | 515 | 28.2 | 93.8 | (91.7–95.9) | 8.7 | (6.2–11.1) | 32.6 | (28.6–36.7) | 1.027 | 0.718 |
| 65+ | 440 | 15.0 | 86.4 | (83.2–89.6) | 11.2 | (8.3–14.2) | 22.5 | (18.6–26.4) | 0.973 | 1.214 |
| All | 2,234 | 37.4 | 91.4 | (90.2–92.5) | 9.8 | (7.3–12.2) | 41.2 | (39.2–43.4) | 1.109 | 0.490 |
| Period 3 (October 2019–February 2020) | ||||||||||
| 5–9 | 489 | 65,0 | 89.3 | (86.6–92.0) | 13.5 | (10.4–16.5) | 62.8 | (58.5–67.1) | 1.032 | 0.795 |
| 10–19 | 519 | 61.5 | 91.9 | (89.5–94.2) | 8.5 | (6.1–10.9) | 59.7 | (55.5–63.9) | 1.004 | 0,959 |
| 20–44 | 3,044 | 53.9 | 95.1 | (94.4–95.9) | 7.1 | (6.2–8.0) | 54.6 | (52.8–56.3) | 1.024 | 0.690 |
| 45–64 | 1,666 | 38.9 | 93.7 | (92.5–94.8) | 10.0 | (8.6–11.5) | 42.6 | (40.2–44.9) | 1.041 | 0.632 |
| 65+ | 1,094 | 24.0 | 90.5 | (88.7–92.2) | 11.5 | (9.6–13.4) | 30.4 | (27.7–33.2) | 1.023 | 0.827 |
| All | 6,812 | 46.8 | 93.5 | (93.0–94.1) | 9.3 | (8.6–10.0) | 48.7 | (47.6–49.9) | 1.031 | 0.695 |
| Period 4 (March 2020–April 2020) | ||||||||||
| 5–9 | 79 | 63.3 | 94.0 | (88.8–99.2) | 20.7 | (11.8–29.6) | 67.1 | (56.7–77.4) | 1.185 | 0.290 |
| 10–19 | 81 | 50.6 | 95.1 | (90.4–99.8) | 15.0 | (7.2–22.8) | 55.6 | (44.7–66.4) | 1.119 | 0.325 |
| 20–44 | 736 | 34.9 | 92.2 | (90.3–94.2) | 10.7 | (8.4–12.9) | 39.1 | (35.6–42.7) | 1.032 | 0.731 |
| 45–64 | 249 | 21.7 | 92.6 | (89.3–95.8) | 11.3 | (7.4–15.2) | 28.9 | (23.3–34.5) | 1.044 | 0.657 |
| 65+ | 189 | 13.2 | 92.0 | (88.1–95.9) | 15.9 | (10.6–21.1) | 25.9 | (19.7–32.2) | 1.093 | 0.505 |
| All | 1,334 | 32.0 | 92.7 | (91.3–94.1) | 12.2 | (10.5–14.0) | 38.0 | (35.4–40.6) | 1.057 | 0.593 |
LR: likelihood ratio.
The overall specificity calculated during the pandemic period was 12.2% (95% CI 10.5–14.0) and was higher than the estimate from Period 3 (p = 0.001), but lower than the estimate from March-April 2019 (p < 0.001). The diagnostic accuracy decreased from 48.7 to 38.0% (22% decrease; p < 0.001) in Periods 3 and 4, respectively, this being similar to the accuracy from Period 1 (p = 0.459).
The AUROCs are presented in Fig. 1 and ranged from 0.544 (95% CI 0.533−0.556) to 0.607 (95% CI 0.586−0.628). No significant differences were documented between the pre- and during-pandemic periods (p = 0.855).
DiscussionThis study evaluated the performance of the influenza case definition based on a national and normative influenza cohort. Our results suggest no significant changes in the evaluated parameters before and during the COVID-19 pandemic.
The analysed cohort study has several strengths and includes: (i) influenza virus infection was confirmed by qRT-PCR analysis, which is the gold standard; (ii) the database included cases from all age groups, and (iii) data for influenza A and B viruses were available.
Timely identification of influenza virus infection can assist healthcare providers in determining optimal strategies for preventing or treating influenza, including the use of antiviral drugs. These interventions also reduce the spread of influenza.11
Multiple case definitions for suspected influenza are currently being used worldwide and include, among others, those proposed by the CDC,11 WHO,12 and the GROG.13 All of them perform similarly in detecting laboratory-positive cases, and their sensitivity and specificity range from 90 to 96% and from 7 to 21%, respectively.5 The AUROCs calculated using any of these classifications are similar to those that were estimated in our study (≈0.550).
No differences in the performance of the case definition or the symptoms in influenza cases according to virus type were found in a recently published study.14 The identified influenza virus sub-types were as follows (n = 7955): A/H1N1, 57%; B/Victoria, 17%; A/H3, 16%; B/Yamagata, 9% and B unidentified, 1%.
Seasonal influenza vaccination has been proven to be cost-effective in terms of prevention.15 However, low acceptance rates have been documented among Mexicans, even in high-risk groups (elderly, about 56%).16 Vaccination coverage among individuals of working age is even lower (20%).17
The potential limitations of the study must be cited. During March 2020, nearly 16% of the cases that met ILL/SARI criteria were confirmed (qRT-PCR) as COVID-19 due to SARS-CoV-2. This proportion reached nearly 50% by April 2020 as it grew with the epidemic. However, we consider that our findings may still be valid for identifying cases that may benefit from the use of neuraminidase inhibitors since the influenza-related burden in Mexico remains high.18
ConclusionsOur findings suggest that the case definition for suspected cases used in community surveillance of influenza has a good performance, even during the COVID-19 pandemic. Therefore, this definition may be used for identifying patients who may benefit from early access to neuraminidase inhibitors. Timely use of antiviral drugs, together with immunization promotion, may reduce the social and economic burden of influenza.
Conflict of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Murillo-Zamora E, Hernández-Suárez C.M. Desempeño de la definición de caso sospechoso de influenza antes y durante la pandemia por COVID-19. Rev Clin Esp. 2021;221:582–586.